|
1/15/2024 0 Comments All vsepr shapes
For the purposes of the VSEPR model, a double or triple bond is no different in terms of repulsion than a single bond. Molecules can be systematically classified according to the number of bonding pairs of electrons as well as the number of nonbonding, or lone pairs, around the central atom. The electron-pair geometries shown in Figure 17.3 describe all regions where. VSEPR structures like this one are often drawn using the wedge and dash notation, in which solid lines represent bonds in the plane of the page, solid wedges represent bonds coming up out of the plane, and dashed lines represent bonds going down into the plane. VSEPR theory predicts the three-dimensional arrangement of atoms in a molecule. .png)
Its tempting to say that the bond angle will be 120 degrees, but bond angles in molecules with lone pairs on the central atom have been observed to be smaller than expected. 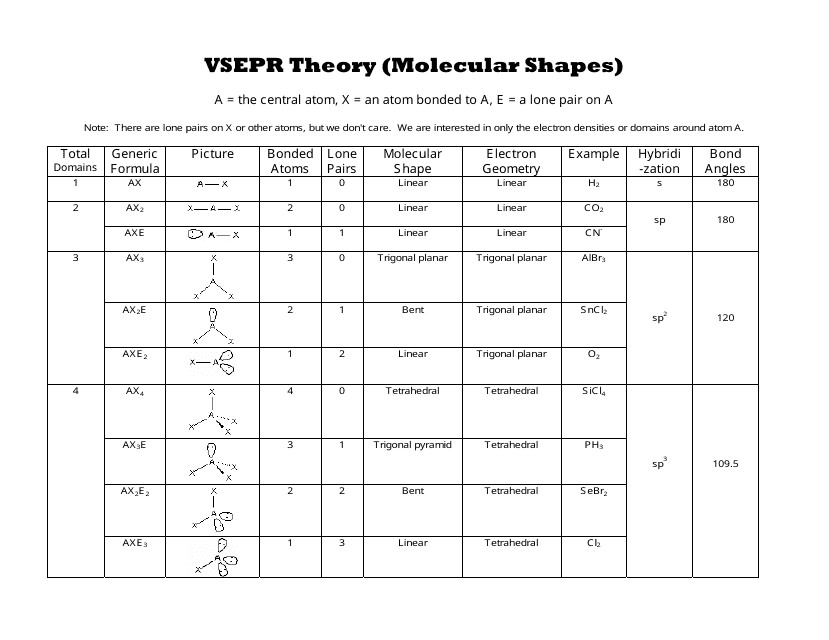
The model states that electron pairs will repel each other such that the shape of the molecule will adjust, so that the valence electron-pairs stay as far apart from one another as possible. The molecular structure of the methane molecule, CH4, is shown with a tetrahedral arrangement of the hydrogen atoms. So, if we were to name this shape, we would focus on the shape that is determined by the atoms. The acronym VSEPR stands for the valence-shell electron pair repulsion model. The valence shell holds the electrons that are involved in bonding and are the electrons shown in a Lewis structure. The valence shell is the outermost electron-occupied shell of an atom. They developed a new approach based on earlier work by other scientists that incorporated a consideration of electron pairs in predicting three-dimensional structure. The problem was that this theory gave an incorrect prediction of bond angles for many compounds. Textbooks(1-3) and online resources(4-5) teach that it is first necessary to draw a Lewis structure before determining the VSEPR shape. The theory at that time relied on hybrid orbitals to explain all aspects of bonding. Depending on how many of the clouds are lone pairs, the molecular geometry will be tetrahedral (no lone pairs), trigonal. To minimize repulsions, four electron clouds will always adopt a tetrahedral electron geometry. Nyholm recognized that the current model for explaining bond angles did not work well. In this video, we apply VSEPR theory to molecules and ions with four groups or clouds of electrons around the central atom. Dipoles and Electrostatic Surfaces XeF4, ClF3 and CCl3Br VSEPR CH4 Methane VSEPR ClF3 Chlorine Trifluoride VSEPR H2O Water VSEPR NH3 Ammonia VSEPR PF5.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
